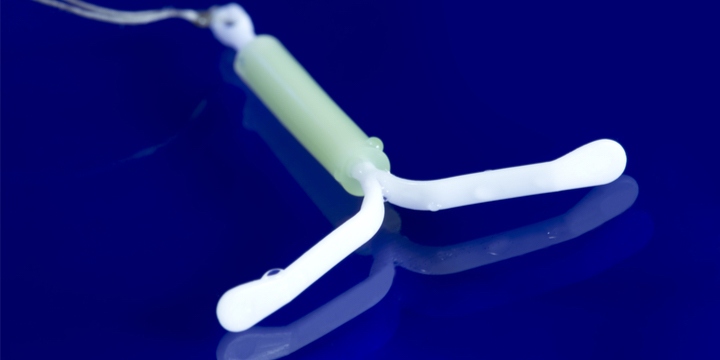
Certain IUDs of the Ancora and Novaplus brands, the marketing of which has already been suspended since November 2019, must be withdrawn preventively, “without urgency”, in particular because of a risk of spontaneous expulsion, the Medicines Agency said on July 27. (ANSM).
This decision could concern 40,000 women in France, carriers of IUDs of these two brands of the Spanish manufacturer Eurogine. They were installed mainly in 2017 and 2018, and “until March 2019”, specifies the ANSM.
“It is a precautionary measure, without urgency, to be foreseen during the next consultation with your gynecologist.“, his general practitioner or his midwife, told AFP Thierry Thomas, deputy director of the ANSM in charge of medical devices.
Read also: Five questions about hormonal and copper IUDs
What signs should alert?
In contrast, “in case of suggestive signs“expulsion (abdominal pain, bleeding out of rules, absent or too long traction thread, pain during sexual intercourse …), the women concerned must have”the reflex to go to consult immediately“, he clarified.
The main risk associated with this lack of stability is the ineffectiveness of the IUD as a means of contraception, which can lead to unwanted pregnancies.
A defective IUD
A recommendation had already been made to remove these IUDs after three years, as the majority of reported expulsions occurred after that time. But the ANSM now recommends removing the intrauterine devices (IUD) concerned without waiting three years.
Indeed, “numerous reports, in particular of pregnancy or spontaneous expulsion, continue to be reported to the ANSM concerning Ancora or Novaplus models having been placed until March 2019“, explains the policeman of health products.
Old IUDs
March 2019 corresponds to the last recall of the affected products implemented by their French distributor, so it is “the last possible date when IUDs could have been inserted“concerned, underlines Thierry Thomas.
This communication is also an opportunity to remind those who have had this model for more than three years but have not yet withdrawn the recommendation.
Remove with care
Due to an increased risk of breakage during removal, healthcare professionals responsible for gynecological monitoring (gynecologists, general practitioners and midwives) are advised to proceed with caution.
“Pull slowly and constantly while pulling the wires, then visually check the integrity of the device once removed. (…) In the event of a rupture and the persistence of a fragment inside the uterus, perform an ultrasound after the next menstruation (the residual fragment can be expelled during menstruation)“, explains in particular the document detailing the recommendations.
Eurogine represented “around 5% of IUD sales in France until 2017“, or some 20,000 devices per year, a proportion which then fell to 3% in 2018, said Thierry Thomas.
 Cherry tomatoes contaminated with salmonella: 92 sick and 1 dead
Cherry tomatoes contaminated with salmonella: 92 sick and 1 dead  A better coaching method can make a person grow
A better coaching method can make a person grow  What is the method to prevent diabetes in children?
What is the method to prevent diabetes in children?  What are the effective factors in causing stomach ulcers?
What are the effective factors in causing stomach ulcers?  Why do embarrassing memories seem to appear at night?
Why do embarrassing memories seem to appear at night?  The amazing link between SARS-CoV-2 infection and newly started diabetes
The amazing link between SARS-CoV-2 infection and newly started diabetes  WHO says monkey pox is not a global emergency right now
WHO says monkey pox is not a global emergency right now  Single cell RNA sequencing uncovers new mechanisms of heart disease
Single cell RNA sequencing uncovers new mechanisms of heart disease  Hepatitis of unknown origin: 3 new deaths and 228 cases worldwide
Hepatitis of unknown origin: 3 new deaths and 228 cases worldwide