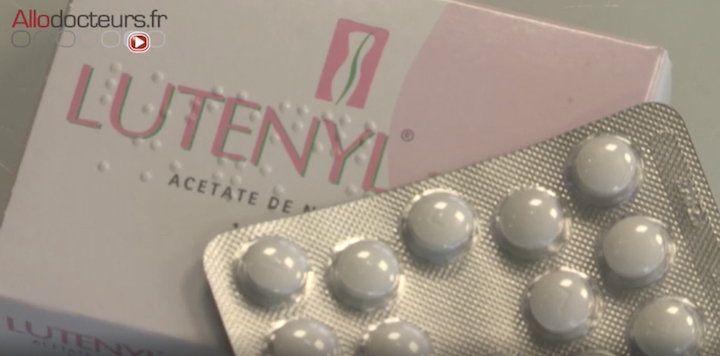
The Medicines Agency (ANSM) has decided to severely restrict the indications for two progestogen treatments, Lutéran and Lutényl. They increase the risk of meningiomas in patients, which are usually non-cancerous brain tumors.
By The editorial staff of Allodocteurs.fr
Written on
Lutéran, Lutenyl and the risk of brain tumors: limited indications
For more than 10 years, Magali, 53, took Lutéran and Lutényl. In 2016, she had a serious epileptic seizure and had to go to the emergency room. “I had a CT scan and very quickly the doctor told me it was meningioma. There was one on the left and another above the right ear. ” These tumors compress his brain and cause edemas, which is responsible for epilepsy. They must be removed, but interventions are risky. “There was a risk of loss of mobility on the right side, arm and leg. And a risk in terms of language. For the second operation, there was another risk which was visual loss on the left side ”.
Endometriosis, functional hemorrhages … more restricted indications
Finally, the operations went well but Magali could not work for a year. And above all, all these health problems could have been avoided. These progestin-only treatments were taken by more than 400,000 women in 2019 for menstrual or menopausal disorders and endometriosis. Magali is however one of the many patients for whom they were not justified.
The Medicines Agency wants to put an end to these abuses and even restrict the initial indications. Dr Isabelle Yoldjian, head of the Gynecological department at ANSM, details the remaining indications: it mainly concerns “endometriosis, functional bleeding such as fibroids, and extremely disabling breast pain. It is in these very specific indications, which are in fact relatively severe, that these treatments can continue to be used ”. The ANSM also requests regular monitoring of women undergoing MRI treatment.
 Cherry tomatoes contaminated with salmonella: 92 sick and 1 dead
Cherry tomatoes contaminated with salmonella: 92 sick and 1 dead  A better coaching method can make a person grow
A better coaching method can make a person grow  What is the method to prevent diabetes in children?
What is the method to prevent diabetes in children?  What are the effective factors in causing stomach ulcers?
What are the effective factors in causing stomach ulcers?  Why do embarrassing memories seem to appear at night?
Why do embarrassing memories seem to appear at night?  The amazing link between SARS-CoV-2 infection and newly started diabetes
The amazing link between SARS-CoV-2 infection and newly started diabetes  WHO says monkey pox is not a global emergency right now
WHO says monkey pox is not a global emergency right now  Single cell RNA sequencing uncovers new mechanisms of heart disease
Single cell RNA sequencing uncovers new mechanisms of heart disease  Hepatitis of unknown origin: 3 new deaths and 228 cases worldwide
Hepatitis of unknown origin: 3 new deaths and 228 cases worldwide